LUNG CANCER AWARENESS MOVEMENT COALITION
Acting locally. Impacting globally.
Our Mission
Bringing together a global coalition to transform lung cancer survival by amplifying the conversation and the importance of early detection, treatment and research.

Building public support for more research, more screening and better treatment options for people with lung cancer.
Building partnerships around the world with patient advocacy organizations, health care NGOs and national ministries of health.
Raising awareness with health care professionals and the general public about how early detection can increase lung cancer survival.
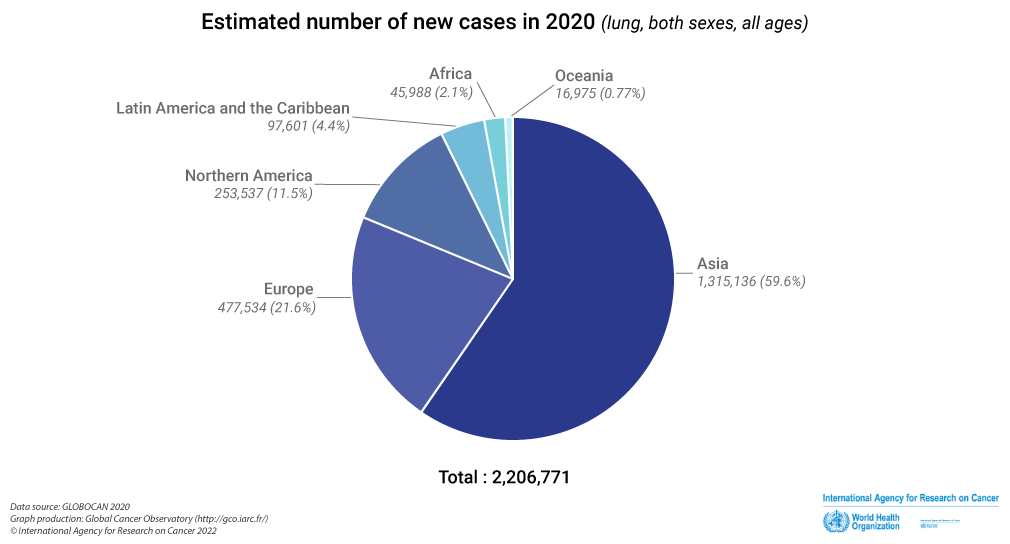
Over 2.2 million people worldwide will get lung cancer annually
Lung cancer kills more people each year than any other cancer
Lung cancer screening saves lives
More research = more treatments = better outcomes
Lung Cancer Around the World
How you can help
Partner
Become part of a global network of organizations working together to raise awareness.
Share the Facts
Share facts about lung cancer to raise awareness in your community.
Advocate
Be an advocate for more funding, more research and more treatment options.
